New regulations passed yesterday define how New Zealand companies can grow, manufacture and sell medicinal cannabis. We spoke to three advocates of medicinal cannabis about their hopes for this emerging industry and how it will affect their lives.
Mention cannabis and the question on everyone’s lips is “are you going to vote to make weed legal?” Most people are thinking about the 2020 New Zealand Cannabis Referendum, where at the election next year we’ll all get to vote on whether recreational weed should be legal.
But there are cannabis-related law changes that were revealed yesterday that could transform lives. The Ministry of Health’s new Medicinal Cannabis Scheme addresses how we cultivate, manufacture and distribute medicinal cannabis on a domestic level.
Basically, New Zealand companies will be able to grow cannabis for the purposes of making medicine. They’ll also be allowed to produce and distribute that medicine.
It doesn’t mean that the weed plant in your backyard is now legal as long you call it “medicinal” - growing or producing cannabis for recreational use is definitely still illegal. And all producers of medicinal cannabis will need a licence. Approved products include tablets, dried products and liquids, but not anything that can be smoked. Products made from synthetic cannabinoids are not included.
The products will still need to be prescribed by a doctor, and can only be bought directly from a medical practitioner or pharmacy - so you won’t be able to buy CBD oil from the supermarket.
The government hopes this will make medicinal cannabis easier and cheaper to access for Kiwis who need it. We spoke to medicinal cannabis advocates to understand how one of New Zealand’s newest industries might take shape.
The Patient – “I have a right to talk about, understand and consent to the medications I put in my body.”
For 23-year-old Christchurch woman Kerri King, it’s a Russian Roulette every time she drinks a glass of water – will it hydrate her or make her vomit?
She suffers from a rare condition called gastroparesis - a partial paralysis of the stomach meaning her muscles don’t function enough to properly digest food. After almost every meal (and she’s been advised to eat six small ones a day), Kerri will experience pain, bloating and nausea.
On top of that she also has extreme muscle pain and fatigue caused by fibromyalgia.
“I have a lot of pain in my arms… people touching me can be painful, even if it’s a light touch,” she explains.

Kerri King
While trying to make the most of her 20s, care for her four-year-old daughter, work, and study part-time Kerri is ‘biting through’ the pain because the tramadol she’s been prescribed only aggravates her gastric symptoms.
“The key point is opiates – like codeine, morphine, tramadol, your stronger pain relievers – slow the stomach. They stop the stomach from emptying and with that already being an issue it can make it worse.”
Kerri has tried recommended medications as well as changing her diet and having botox injected into the bottom of her stomach.
“Botox has like a 40-50% success rate and it has to be redone every 12 weeks,” she says, “For me it only lasted six weeks before it stopped working.”
There is however one treatment that Kerri favours, but up until now it’s been almost impossible to get.
“In the first instance, it was trying medicinal cannabis illegally,” she says.
“And then because I couldn’t get my GP to even talk to me about it, I searched up one of the cannabis doctors in Auckland… and eventually got a script to try (legally)”.
When we’re talking about cannabis, there are two main substances to know about. One is tetrahydrocannabinol, commonly known as THC, which is a psychoactive that gets you high. The other is cannabidiol, commonly known as CBD, which is a compound with potential therapeutic benefits that has little to no psychoactive effect.
Kerri’s first prescription was for 50 days worth of Tilray cbd100, which is a CBD oil with zero THC.
Until now, regular GPs were able to prescribe cannabis medicines with no more than 2% THC (the cannabinoid associated with euphoria). For anything with higher levels of THC sign-off from a specialist and ministerial approval was required.
A recent report by Royal Society Te Apārangi found evidence that cannabis-based medicines have health benefits in certain situations including chronic pain, chemotherapy induced nausea, muscle spasms due to multiple sclerosis and certain types of epilepsy. At the same time, the report identified large gaps in understanding the health effects of cannabis, saying we need to know more about the effects on population groups thought to be most at risk from cannabis use: those under 18 years, pregnant and breastfeeding women and heavy users.
When Kerri tried to renew her script in Christchurch, she was served a flat ‘no’’ on the grounds her regular GP clinic wanted to stay within their scope of practice and there was not enough evidence of the medicine’s efficacy.
“Medical Cannabis Awareness New Zealand was happy to educate them about it and they weren’t having a bar of it,” she says.
When asked why she wouldn’t just change practices Kerri says, “Continuity of care is important. A doctor who knows your health history should be able to make better choices around what you need and what will work.
“If you’re constantly hopping around doctors, you’re having to spend that 15-minute appointment re-explaining your entire thing.
“I’ve already spent a lot of time educating doctors that I’ve been seeing within my practice about my condition so if I have to change that process just keeps starting all over again."
New regulations will dictate what a new prescribing process may look like and some GPs are hoping they may be able to forego some of the additional approvals currently required.
This could be potentially life-changing for Kerri but she says if doctors aren’t educated and wait times remain the way they are then a law change isn’t enough.
“Our medical system is just ridiculous to navigate. It takes a very long time. My first appointment, I waited five months for, for them to tell me nothing was wrong. I ended up in hospital two weeks later, it was just unreal.”
“Now I feel like I know more about my conditions than my GP does and they will acknowledge that but yet they won’t listen to me about the choices I’m making around the medications I want to treat my conditions.”
The Doctor – “No doctor likes to prescribe second-best”
As part of the new changes, a medicinal cannabis agency has been set up under the Ministry of Health that will be up and running from 1 April 2020. They’ll issue the licenses that allow manufacturers to operate legally, with the first licenses likely to be given from mid-way through next year. They also will administer quality standards, which include things like batch testing to measure the level of active ingredients, shelf life, and to check for contamination.
Dr Graham Gulbransen, who has been a GP for 35 years, an addictions specialist for 10 years and a cannabis consultant for the last two years, hopes affordability and accessibility for patients like Kerri will improve.

Dr Graham Gulbransen
“As a doctor I can prescribe very strong drugs like opioids for pain or anticoagulants or insulin for diabetes but when it comes to THC to give additional pain relief, we’ve [had] this difficult process,” says Dr Gulbransen.
Dr Gulbransen has directly prescribed CBD oil to about 250 patients since 2017, when CBD was changed from a controlled drug to a drug falling within the Medicines Act. In addition he has supported several applications for medicines with higher levels of THC.
“Some patients think it’s a bit of a con that they come to me as a cannabis specialist but if they want something with THC they have to have a form signed by another specialist as well.”
He says he understands why doctors may be conservative about prescribing the drug in cases like Kerri’s (who Dr Gulbransen has not consulted on).
“There’s a lot of resistance… they would like to know that these are safe and have been tested and I think the New Zealand industry is going to have to follow those processes to assure doctors that their products are safe to prescribe.”
He thinks people can be resistant to medicinal cannabis because they associate it with recreational.
“In a way we need to separate recreational or adult use from medicinal use and there is some data on the risk of addiction with adult use and it suggests that perhaps 10-15% of people get into trouble from misusing cannabis,” he explains.
“Usually those people have underlying problems of childhood abuse or anxiety and depression and they may be self-medicating with cannabis.”
Dr Gulbransen also points out the risk for dependence on opioids – which are currently the easiest prescriptible methods for pain relief – is widely known and evidenced.
Dr Gulbransen surveyed the first 200 patients he prescribed CBD oil to and found 48% of patients with chronic pain reported very good to excellent results with some able to reduce or stop using opioids. His findings will soon be published in the British Journal of General Medicine.
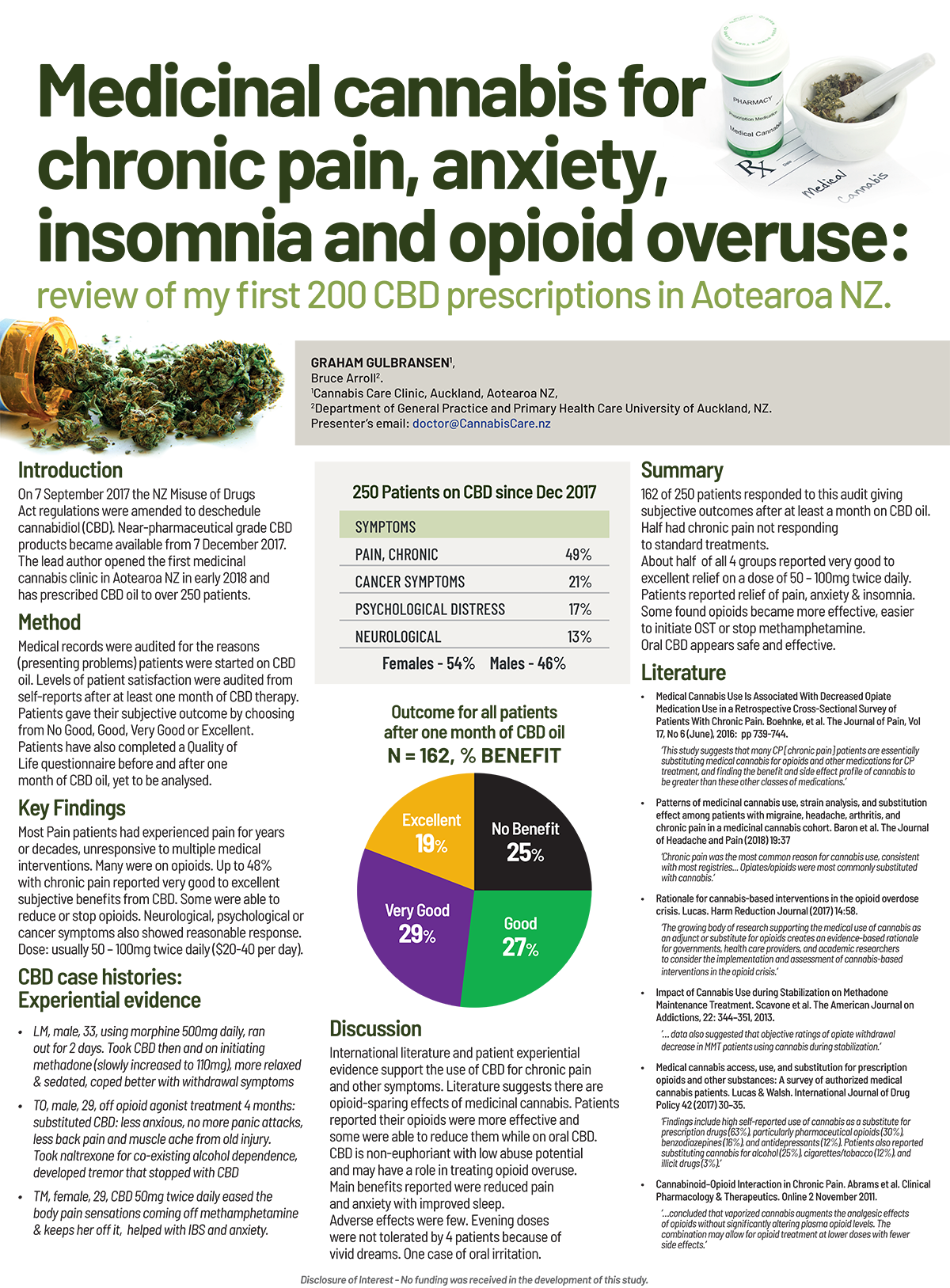
Dr Gulbransen's findings are soon to appear in the British Journal Of General Medicine
However, even if patients get access to the medication, the high cost of it can be prohibitive. Up until yesterday’s changes, all CBD treatment had to be imported from overseas. The main medicines (Tilray and Sativex) come from Canada and Switzerland respectively. The costs to Dr Gulbransen’s patients have been between $20 - $40 a day.
In his announcement of the law changes on Wednesday, the Minister of Health Dr David Clark said he expected New Zealand companies would be able to lower the cost. “I’m confident increased competition from local manufacturers will drive prices down over time,” Dr Clark said in a press release.
Dr Graham Gulbransen is hopeful for a significant change to the way things are now.
“People are going to keep smoking cannabis. They’ve used cannabis for 10,000 years and I think it would be much safer if people were able to go to a regulated outlet and buy a standardised product and know what was in it and know that it had been tested for quality and safety.”
The Corporate – “We don’t view ourselves as a ‘big corporate’”
There’s already 20 businesses in New Zealand with a license to grow cannabis for research purposes. And 238 are growing industrial hemp (a strain of cannabis with low levels of THC, the psychoactive in weed that gets you high). These companies will now be able to apply for licenses to pivot from research or hemp to cannabis.
As well as needing licenses and testing, producers will be required to have secure premises and will have to pay a fee to the government which will help cover the cost of administering the new regulation.
There’s an opportunity for corporate medicinal cannabis companies to thrive in New Zealand and abroad but there are questions over whether the to-be established industry will be less about patient care and more of a cash-grab.
Aldo Miccio, former mayor of Nelson and chairperson of Medical Kiwi New Zealand, a medicinal cannabis company based in the South Island says that’s not the case.
“We don’t view ourselves as a big corporate… We’re a health and wellness company and we want to operate with medicine, the pharmaceutical products and we also want to operate with wellness products.
“They’re both very different, but they both happen to be from the cannabis plant.”

Artist's impression of the Medical Kiwi Facility
Medical Kiwi’s proposed $15 million Nelson-based facility is set to be one of the biggest medicinal cannabis research and distribution centres in the country. Earlier this year they were the first South Island company to be granted a cultivation licence which means now that regulations are set, they’re ready to begin operations.
It’s hoped locally cultivated cannabis will mean more range to choose from at cheaper prices for patients. But even now that the changes to regulation allow for this to happen, Aldo anticipates it will take Medical Kiwi 12 to 18 months to produce any pharmaceutical products for patients to use.
Medicinal cannabis advocates like Aldo had hoped for a possible CBD-based ‘wellness’ industry, where any Kiwi could buy topical balms or oral sprays as easily as we can buy St. Johns Wort at the local pharmacy.
Aldo puts a lot of emphasis on this proposed ‘wellness arm’ of Medical Kiwi. He had hoped that ‘food grade’ CBD products - like topical balms, sprays and tinctures - would be able to be sold liberally while the pharmaceutical side of the business is getting off the ground.
But the regulation change hasn’t delivered on this - you’ll still need a doctor’s prescription, and you won’t be able to get it from the supermarket.

Vitality oral drops and spray
“CBD doesn’t have the psychoactive effects that THC has, it’s been a food product for thousands of years, other countries consider it a food product and it has great health benefits, nutraceutical health benefits, so there’s no reason why it shouldn’t be non-prescription,” says Aldo.
It’s for that reason Medical Kiwi has been forging relationships within established markets overseas – one of those is Vitality CBD based in the UK.
Liam Cagliarini, sales director for Vitality, uses CBD personally for muscle recovery and to help with sleep.
“In the UK they’ve actually classified CBD as being a food supplement so therefore it’s not a medicinal product, you can’t make any medical claims but the benefit for the industry is that CBD is in massive consumer demand.”
Vitality’s products are advertised to help with sleep, anxiety, depression, inflammation and pain relief. They derive their CBD from the hemp plant which means there’s little to no THC from the outset – it doesn’t have to be separated like other cannabis medicines.

“Cannabis is a family,” explains Liam.
“The best analogy I can give this is on the left hand side you have marijuana which is like the naughty brother which has a high level of THC.
“On the right hand side you have hemp which has high amounts of CBD and low amounts of THC and that’s where we extract the CBD from.”
CBD from the hemp plant currently falls under prescription and companies like Medical Kiwi need a cultivation license to import and distribute it.
Dr Gulbransen says there is some benefit to establishing a ‘food grade’ market alongside pharmaceutical grade.
“I think food grade might be less stringent in its quality and patients would take it as a supplement in a way that they might buy other herbal supplements such as St. John’s Wort."
But how do we know food grade CBD isn’t another gimmicky product that we could buy from a store that smells a little too strongly of incense?
“It’s a health product, it’s a wellness product, it shouldn’t look gimmicky,” explains Liam.
“We publish all our testing… If you look at the New Zealand market, there’s a lot of players on the black market so people making it in their kitchens, selling it out of the boot of their car. Straight away as the consumer you don’t know exactly what you’re taking.”
Tests are done by a third party and published on their website.
“We test the seed, we test for precious metals, pesticides obviously, THC and then we also test when it’s shelf-ready as well.”
At this stage hopes of getting these products on supermarket shelves are simply that – hopes. Regardless of whether that happens, Medical Kiwi will still be forging ahead with the development of the pharmaceutical arm of the company, which will take some time.
Medical Kiwi and other medicinal cannabis companies have been waiting on the regulations to be announced for a year. With cost and accessibility being at the forefront of concerns, Aldo claims once a pharmaceutical products becomes available, it will be just as cost-effective as the current green fairy market.
“The reality is pricing is coming down as more countries deregulate and in the space we’re at, we believe we will be at least a quarter of that price ($600 - $800+ per month) in terms of the medicinal product with THC.
“We will aim to be at the current – you know, green fairy market – we will not be more expensive than the existing green fairy market.”
The future – “People will learn to trust it”
Whether you agree cannabis should be corporatised or not, regulations announced this week allows the industry to put down their roots.
Currently cannabis as a medicine is widely sought after, but is expensive and very hard to access. Both Aldo and Dr Gulbransen cite prohibition as the reason this has been so hard and slow-moving.
Aldo concludes, “I know out in the marketplace there’s a lot of thinking about ‘cannabis is bad’ which yes it is, but when you use it for medicine, it’s just like opium – when used for medicine it’s good and that’s accepted.”
“Once it’s regulated and produced under pharmaceutical grade conditions, people will learn to trust it as a medicine as opposed to seeing it as a recreational drug.”